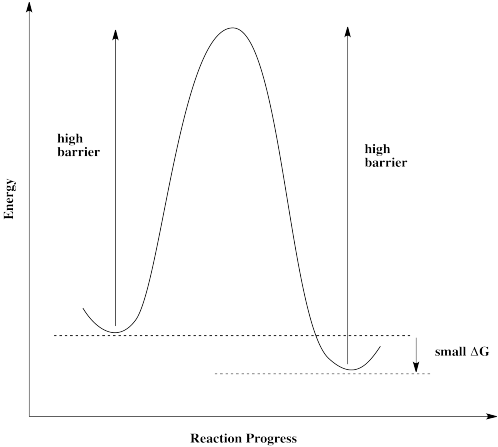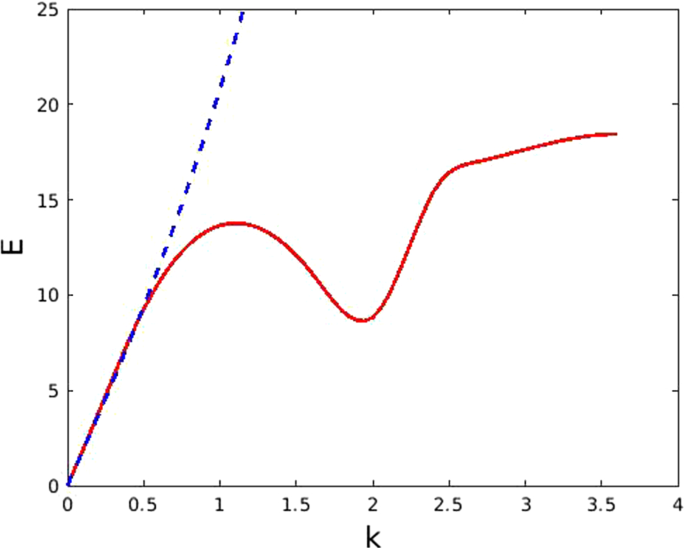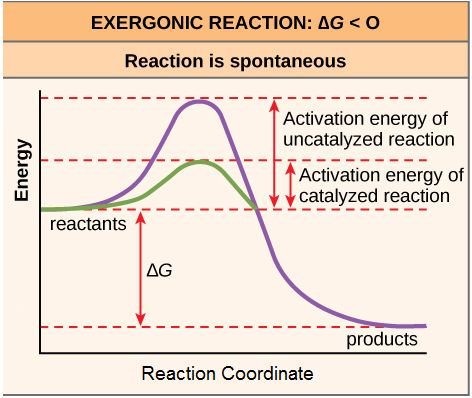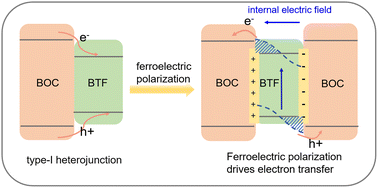
Breaking through the interfacial energy barrier limitations of type-I heterojunctions via ferroelectric polarization engineering: a case study of Bi5Ti3FeO15/BiOCl - Inorganic Chemistry Frontiers (RSC Publishing)

Measuring the Energy Barrier of the Structural Change That Initiates Amyloid Formation | Analytical Chemistry
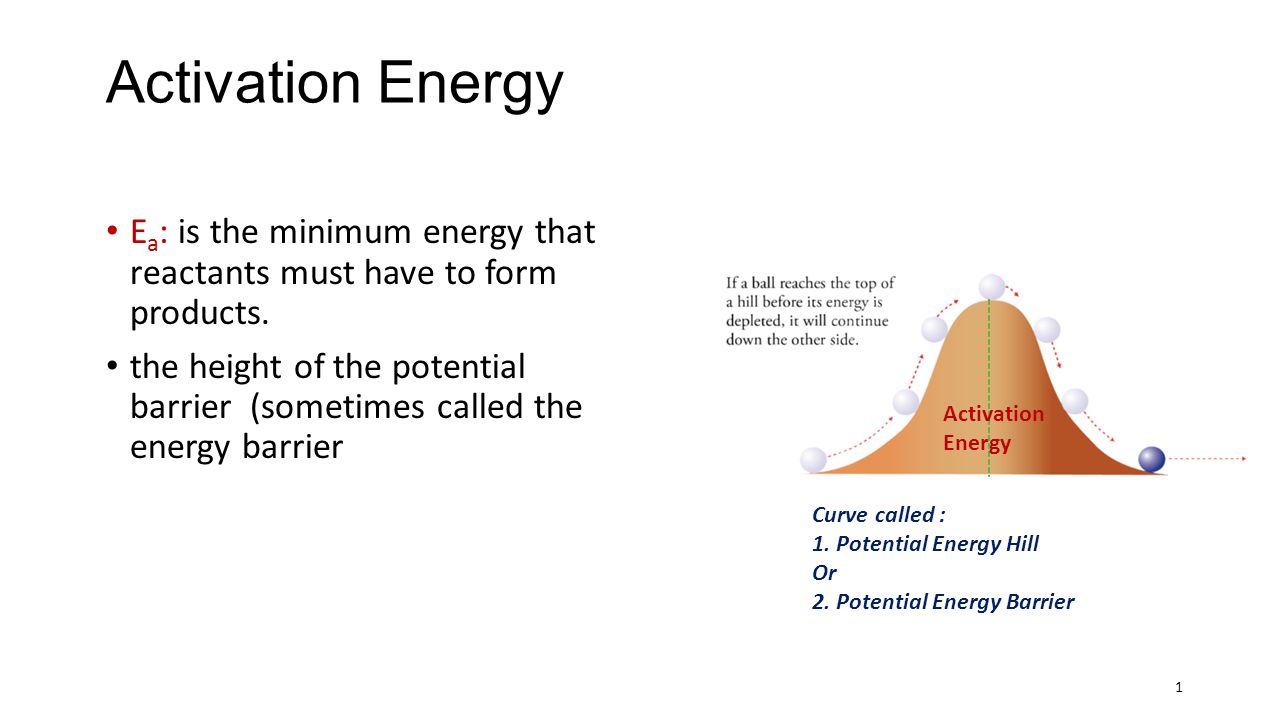
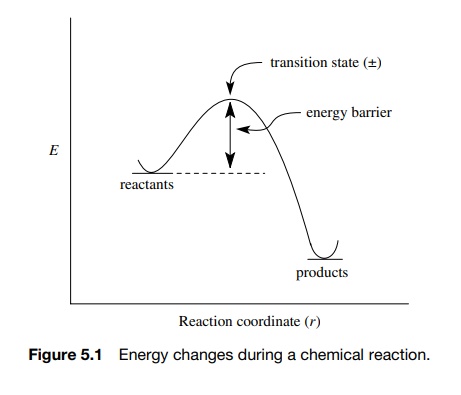
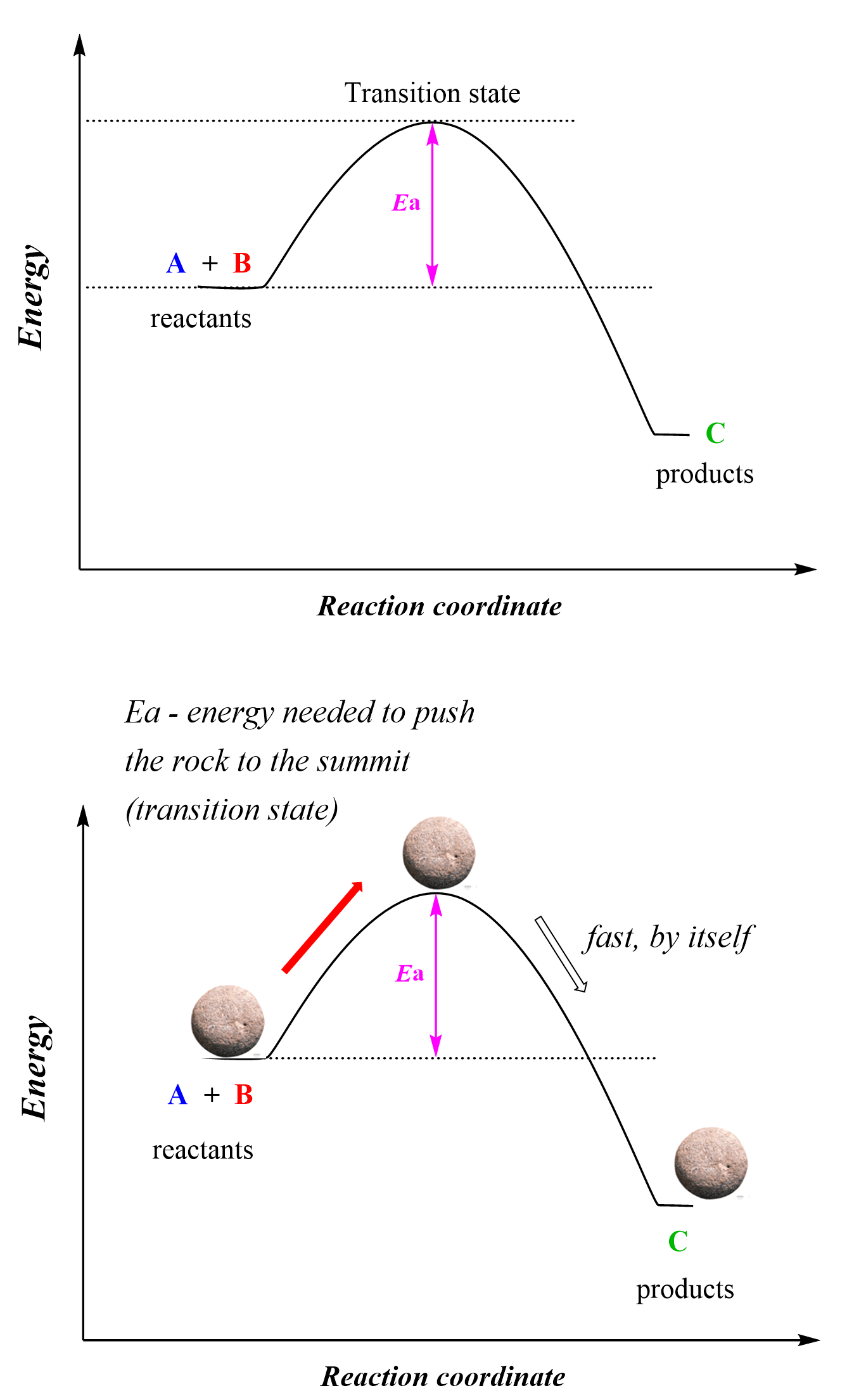
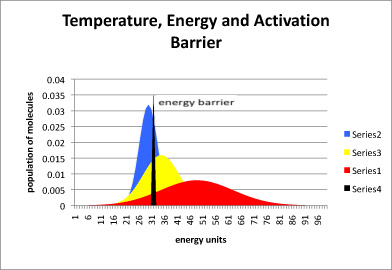
Activation Energy E a : is the minimum energy that reactants must have to form products. the height of the potential barrier (sometimes called the energy. - ppt download

Symmetrical energy barrier for a flow process. The height of the energy... | Download Scientific Diagram
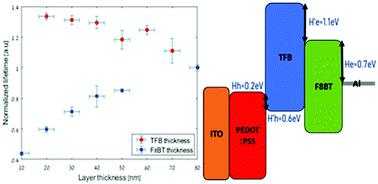
The influence of the internal interface energy barrier and the device dimensions on the transient electroluminescence lifetime of bi-layer OLEDs - Journal of Materials Chemistry C (RSC Publishing)

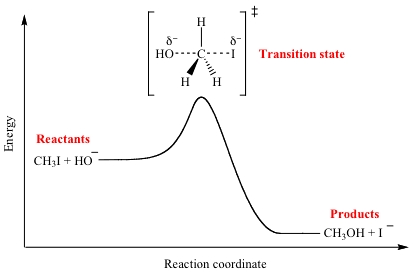
SOLVED: Energy of Activation. The energy barrier (the hump) between the reactants and the products is called the energy of activation, or Ea (Figure 6.13). This energy barrier represents the minimum amount

What's the height energy barrier to convert a gauche to its anti conformation? (the answer key says 5Kcal/mol - 0.6kcal mol = 4.4 kcal/ mol) But I don't understand why! Isn't eclipsed